複素環化合物の効率的合成法の開発
メンバー: 齊藤亜紀夫
分野: 基礎化学、薬学
所属: 工学研究院応用化学部門
キーワード: 複素環、触媒反応、連続反応、多成分反応、ヨウ素
ウェブサイト:
研究概要
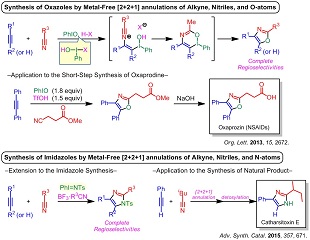
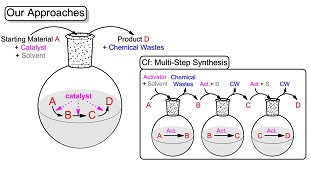
省資源化が求められている現代社会において、有用物質をより効率良く生産するために、「炭素資源の有効活用」や「化学廃棄物の低減化」に配慮した新規かつ効率的な有機分子合成法がいっそう強く求められています。我々は、このような背景のもと、医・農薬や機能性材料などの多くの有用物質に含まれる複素環化合物に注目し、触媒的な連続反応や多成分反応を利用した効率的合成法を開発しています。触媒的な連続反応や多成分反応は、単一の触媒系が1つのフラスコ内で複数の反応を連続的に活性化するので、「操作・処理が簡便」になるだけでなく、「原子効率性の向上」や「化学廃棄物の軽減」につながる環境調和型合成法となります。また、ヨウ素が日本国内に豊富に存在する元素資源であることから、最近、ヨウ素試薬を用いる合成法を中心に研究しています。下記の例はこれまでに開発してきた合成法の例です。
1) 超原子価ヨウ素試薬を「活性化剤」兼「ヘテロ原子供与体」とする、アルキンとニトリルとヘテロ原子との[2+2+1]環形成反応(オキサゾールやイミダゾールの合成法)
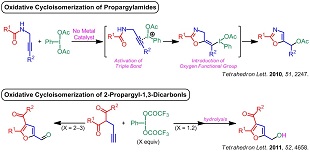
2) 超原子価ヨウ素試薬を「活性化剤」兼「ヘテロ原子供与体」とする酸化的環化異性化反応(側鎖に酸素官能基の導入を伴ったオキサゾールやフランの合成法)
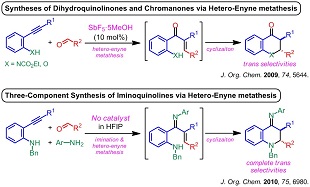
3) ヘテロ-エンインメタセシス反応を経由するジヒドロキノリノンやイミノキノリンの合成法
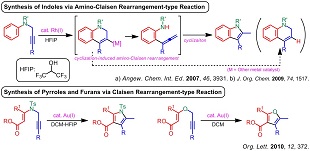
4) アミノクライゼン転位型反応を経由するインドールやピロールの合成法
主要論文・参考事項
(1) "Metal-Free [2+2+1]Annulation of Alkynes, Nitriles and N-Atoms from Iminoiodanes for Synthesis of Highly Substituted Imidazoles", A. Saito, Y. Kambara, T. Yagyu, K. Noguchi, A. Yoshimura, V. V. Zhdankin, Adv. Synth. Catal. 2015, 357, 667-671.
(2) "Metal-Free [2+2+1]Annulation of Alkynes, Nitriles, and Oxygen Atoms: Iodine(III)-Mediated Synthesis of Highly Substituted Oxazoles", A. Saito, A. Taniguchi, Y. Kambara, Y. Hanzawa, Org. Lett. 2013, 15, 2672–2675.
(3) "PIDA-Mediated Synthesis of Oxazoles through Oxidative Cycloisomerization of Propargylamides", A. Saito, A. Matsumoto, Y. Hanzawa, Tetrahedron. Lett. 2010, 51, 2247–2250.
(4) "Synthesis of Pyrroles by Gold(I)-Catalyzed Amino−Claisen Rearrangement of N-Propargyl Enaminone Derivatives", A. Saito, T. Konishi, Y. Hanzawa, Org. Lett. 2010, 12, 372-374.
(5) "Tandem Synthesis of 2,3-Dihydro-4-iminoquinolines via Three-Component Alkyne-Imine Metathesis", A. Saito, J. Kasai, T. Konishi, Y. Hanzawa, J. Org. Chem. 2010, 75, 6980–6982.
(6) "アルキン類の触媒的逐次反応を基盤とする複素環合成法の開発", 齊藤亜紀夫, 榛澤雄二, 有機合成化学協会誌, 2014, 72, 246–256. (account)
お問い合わせ先
東京農工大学・先端産学連携研究推進センター
urac[at]ml.tuat.ac.jp([at]を@に変換してください)
Development of efficient synthetic methods of heterocyclic compounds

Research members: Akio Saito PhD.
Research fields: Basic chemistry, Pharmacy
Departments: Division of Applied Chemistry, Institute of Engineering
Keywords: heterocycles, catalysis, consecutive reaction, multi-component reaction, iodine
Web site:
Summary

Resource-saving technologies are required in the modern society and thus the development of novel and effective procedures for syntheses of useful organic molecules is a crucial issue. From these viewpoints, considering "the effective utilization of carbon resources" and "reduction of chemical wastes", we have developed synthetic methods of heterocycle compounds, which are prevalent in a wide variety of useful compounds (such as medicines, agrichemicals, functional materials and so on), based on catalytic consecutive reactions and multi-component reactions. Since the catalytic system for the catalytic consecutive reactions and multi-component reactions activates plural reactions in one flask, these reactions provides us with not only "simpleness and easiness of operation" but also "improvement of the atom effectiveness" and "reduction of chemical wastes". Recently, we have mainly researched methods with iodine reagents because Japan is abundant in iodine. The following examples are some of our synthetic methods.
1) Syntheses of oxazoles and imidazoles based on [2+2+1]annulations of alkynes, nitriles and heteroatoms mediated by hypervalent iodine reagent as "a activating agent" and "a heteroatom donor"
2) Syntheses of oxazoles and furans concomitantly with incorporation of oxygen functional groups based on the oxidative cycloisomerization of alkynes mediated by hypervalent iodine reagent as "an activating agent" and "a heteroatom donor"
3) Syntheses of dihydroquinolinones and iminoquinolines based on the hetero-enyne metathesis reaction
4) Syntheses of indoles and pyrroles based on the amino-Claisen rearrangement-type reaction
Reference articles and patents
(1) "Metal-Free [2+2+1]Annulation of Alkynes, Nitriles and N-Atoms from Iminoiodanes for Synthesis of Highly Substituted Imidazoles", A. Saito, Y. Kambara, T. Yagyu, K. Noguchi, A. Yoshimura, V. V. Zhdankin, Adv. Synth. Catal. 2015, 357, 667-671.
(2) "Metal-Free [2+2+1]Annulation of Alkynes, Nitriles, and Oxygen Atoms: Iodine(III)-Mediated Synthesis of Highly Substituted Oxazoles", A. Saito, A. Taniguchi, Y. Kambara, Y. Hanzawa, Org. Lett. 2013, 15, 2672–2675.
(3) "PIDA-Mediated Synthesis of Oxazoles through Oxidative Cycloisomerization of Propargylamides", A. Saito, A. Matsumoto, Y. Hanzawa, Tetrahedron. Lett. 2010, 51, 2247–2250.
(4) "Synthesis of Pyrroles by Gold(I)-Catalyzed Amino−Claisen Rearrangement of N-Propargyl Enaminone Derivatives", A. Saito, T. Konishi, Y. Hanzawa, Org. Lett. 2010, 12, 372-374.
(5) "Tandem Synthesis of 2,3-Dihydro-4-iminoquinolines via Three-Component Alkyne-Imine Metathesis", A. Saito, J. Kasai, T. Konishi, Y. Hanzawa, J. Org. Chem. 2010, 75, 6980–6982.
(6) "アルキン類の触媒的逐次反応を基盤とする複素環合成法の開発", 齊藤亜紀夫, 榛澤雄二, 有機合成化学協会誌, 2014, 72, 246–256. (account)
Contact
University Research Administration Center(URAC),
Tokyo University of Agriculture andTechnology
urac[at]ml.tuat.ac.jp
(Please replace [at] with @.)